Search Posts
Recent Posts
- Real Estate in RI: Milk Can Property Churns Up Strong Interest May 15, 2026
- GriefSPEAK: A Stroke of Genius – Mari Nardolillo Dias May 15, 2026
- Outdoors in RI: Quahog Week, Surf Therapy, Be a Lifeguard, Marine Life & Wind Turbines May 15, 2026
- Rhode Island Weather for May 15, 2026 May 15, 2026
- Cranston City Council Begins Reworking Mayor’s Revised Budget After Emotional Hearing – VIDEO May 15, 2026
Categories
Subscribe!
Thanks for subscribing! Please check your email for further instructions.
Desperate times & innovative treatments – Convalescent Plasma for Coronavirus
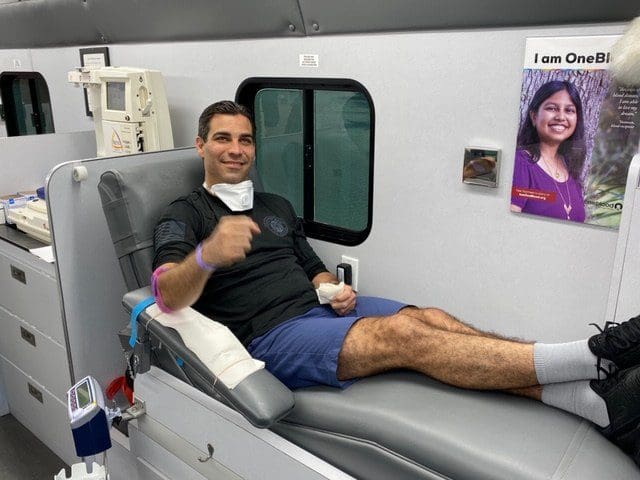
Photo: Mayor Francis X. Suarez, Mayor of Miami, was the first coronavirus survivor to become a plastma donor – his plasma was given to a patient near death – who is still alive and all are tracking his progress.
The FDA has given investigational approval for the use of Convalescent Plasma as an emergency treatment for Covid-19 in very serious cases that are not responding to other treatments and have a dire outcome predicted.
What is convalescent plasma?
• Convalescent plasma is the liquid part of blood that is collected from patients who have recovered from an infection.
• Antibodies present in convalescent plasma are proteins that might help fight the infection. Why is convalescent plasma being investigated to treat COVID-19?
• Convalescent plasma is being investigated for the treatment of COVID-19 because there is no approved treatment for this disease and there is some information that suggests it might help some patients recover from COVID-19.
• Further investigation is still necessary to determine if convalescent plasma might shorten the duration of illness, reduce morbidity, or prevent death associated with COVID-19. Is convalescent plasma safe and effective against COVID-19?
• It is not known if convalescent plasma will be an effective treatment against COVID-19.
• Plasma transfusions are generally safe and well-tolerated by most patients but can cause allergic reactions and other side effects. It is also not known if patients with COVID-19 might have other types of reactions to convalescent plasma.
• While it is not known if convalescent plasma is safe and effective against COVID-19, there is anecdotal evidence that convalescent plasma might be for some patients.
• Because there are no approved treatments, FDA is permitting the emergency investigational use of convalescent plasma to treat COVID-19 under the criteria of the emergency IND.
What is an emergency IND?
A physician may decide to request use of an investigational product through a single-patient Emergency Investigational New Drug (eIND) application if:
• the physician considers the product may be urgently needed for the patient’s serious or immediately life-threatening disease or condition;•no satisfactory alternative therapy is available;
• the probable risk from the investigational drug is not greater than the probable risk from the disease or condition; and
• the patient cannot receive the product through any existing clinical trials or expanded access protocols
Where can a physician obtain convalescent plasma for a patient with COVID-19?
• Convalescent plasma is not yet widely available from blood centers, although many are starting to collect it.
• Hospitals should contact their blood supplier to discuss possible options for obtaining convalescent plasma.
• FDA does not provide convalescent plasma to hospitals. I recently recovered from COVID-19, can I donate convalescent plasma?
• COVID-19 convalescent plasma must only be collected from recovered individuals if they are eligible to donate blood.
• Individuals must have had a prior diagnosis of COVID-19 documented by a laboratory test and meet other laboratory criteria.
• Individuals must have fully recovered from COVID-19, with complete resolution of symptoms for at least 14 days before donation of convalescent plasma. I think I had COVID-19, but I did not get a test. Can I still donate convalescent plasma?
• Yes. Please refer to the information in “Considerations for eligibility of plasma donors” on the investigational COVID-19 convalescent plasma web page. Who should I contact if I want to donate convalescent plasma?
• Not all blood centers collect convalescent plasma, but some have started to collect it.
• You can ask your local blood center if there are options to donate convalescent plasma in your area. FDA included contact information for blood organizations in its March 19, 2020 statement on blood donations.
• FDA does not collect convalescent plasma.
(information courtesy FDA)

